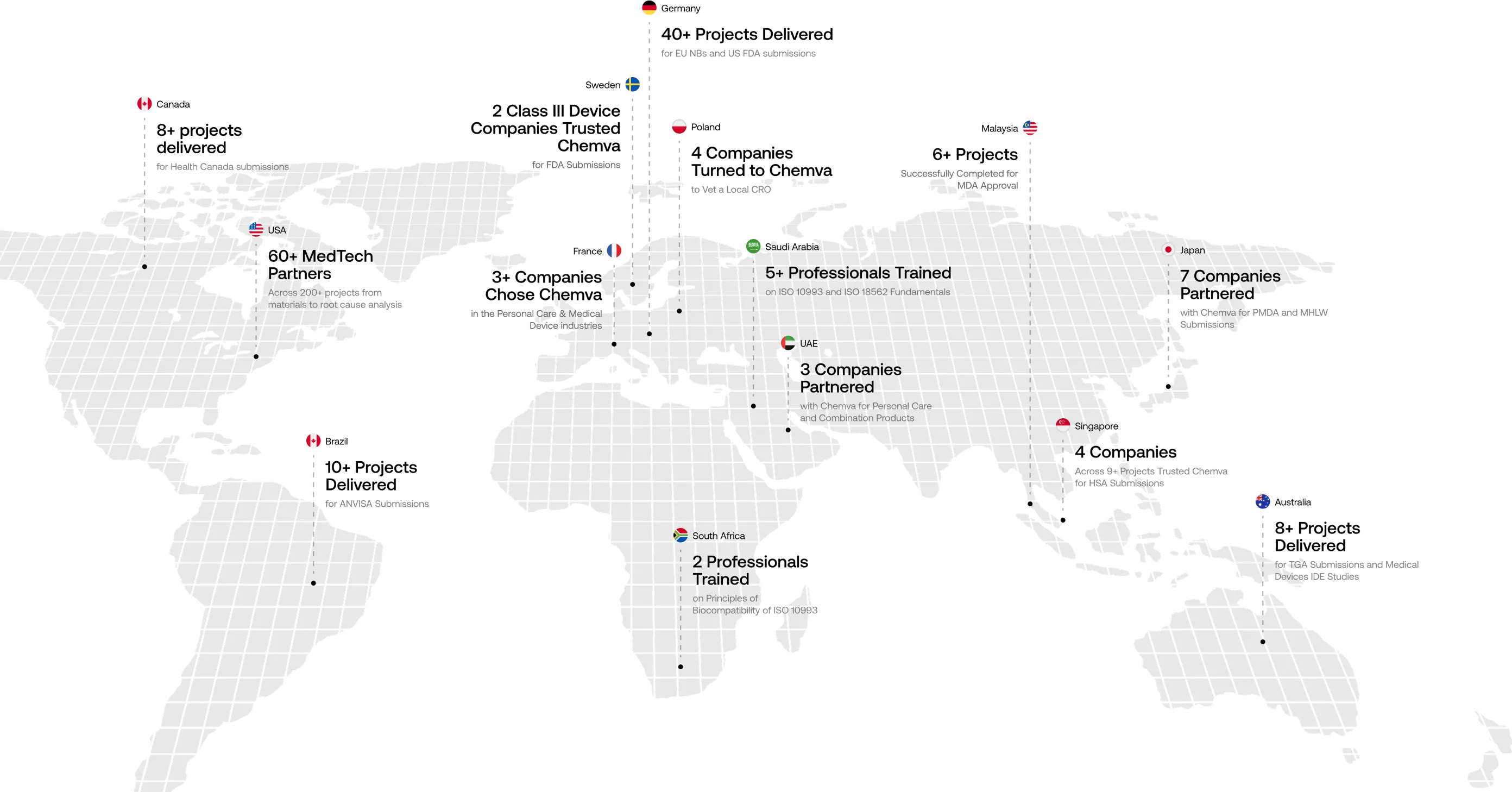
About Chemva
At Chemva, we are more than a consulting firm — we’re a scientific partner
trusted by companies across the globe.

Board Member, Technical
Dr. Rahimi serves as the Technical Board Member at Chemva, where he provides strategic guidance to the company in the consulting areas of biocompatibility, toxicology, chemistry (extractables & leachables), and materials/polymer science. Dr. Rahimi is a recognized expert in the MedTech industry specializes in biological evaluations across all development phases from a global perspective, covering Class I-III devices and combination products, with comprehensive experience working with various notified bodies and national regulatory authorities.Dr. Rahimi holds a B.Sc. in Chemistry, Ph.D. in coatings and polymeric materials, diplomat of the American board of toxicology accreditation (D.A.B.T.), and graduate certificate in statistics. He is a faculty of chemical characterization, materials science, and toxicology with AAMI (the Association for the Advancement of Medical Instrumentation) and co-chairs degradation-related medical device standards at the ISO/AAMI level and represent the US a biocompatibility, E&L, and materials expert.

Biocompatibility Specialist | Materials Engineer
Mr. Taha serves as the Biocompatibility Specialist/Materials Engineer at Chemva, where he supports MedTech companies in the consulting areas of biocomptability, toxicology, chemistry (extractables & leachables), and materials/polymer science. Mr. Taha holds a B.Eng. in Polymer Engineering and has over five years of experience preparing toxicological risk assessments, biological evaluation plans and reports, and change control assessments.

Statistician
Kinza serves as a statistician at Chemva, where he supports MedTech companies in the consulting areas of designing studies and analyzing data to extract informative trends. Kinza holds a B.Sc. and M.Sc. in statistics and has over ten years of experience developing predicitve models for various industiries.

Ashidah serves as a Toxicologist at Chemva, where he supports MedTech companies in the consulting areas of biocompatibility and toxicology. Ashidah holds a M.Sc. in toxicology and has over five years of experience preparing toxicological risk assessments, and biological evaluation plans and reports. She is also an expert locating analogues for toxicological risk assessments.

At Chemva, we are more than a consulting firm — we’re a scientific partner
trusted by companies across the globe.